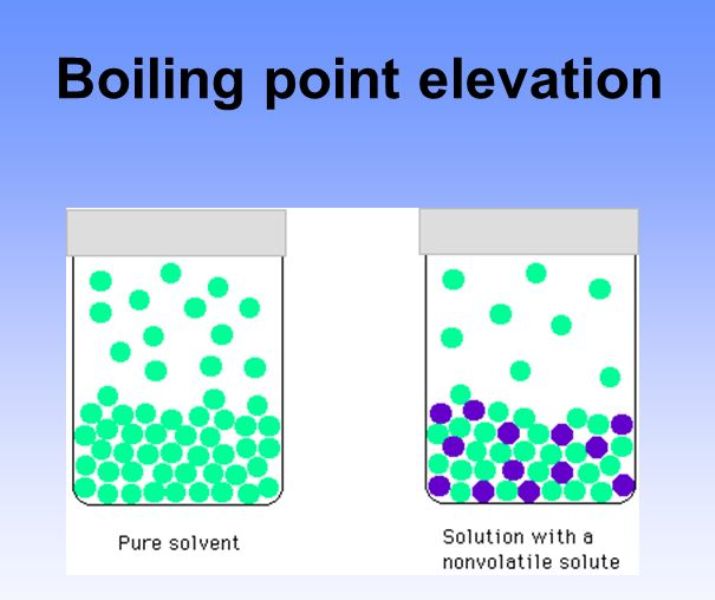
We have more opportunity for London dispersion forces. If I draw in another molecule of hexane, so over here, I'll draw in another one, hexane is a larger hydrocarbon, with more surface area. Transient attractive forces between these two molecules of pentane. So I could represent the London dispersion forces like this. They are attractions between molecules that only exist for a London dispersion forces are the weakest of our intermolecular forces. London dispersion forces, so London dispersion forces exist between these two molecules of pentane. Intermolecular force that exists between two non-polar molecules, that would of course be the Intermolecular forces that exist between those 
Hexane has six carbons, one, two, three, four, five, and six. 
Pentane has five carbons, one, two, three, four, five, so five carbons for pentane. These are both hydrocarbons, which means they contain Pentane on the left and hexane on the right. And those attractionsīetween the molecules are called the intermolecular forces. When its molecules have enough energy to breakįree of the attractions that exist between those molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
